-



-
Small Molecule Drug Platform

-
Chiral Catalysis Technology
-
Continuous Flow Technology
-
Fluorine Chemistry
-
Enzyme Catalysis
-
Photocatalysis
-
Solid-State Research
显示更多 -
-
Drug Product (DP) Platforms

-
Sustained and Controlled Release
-
Solubility Enhancement
-
Topical Formulation
-
Liquid Formulation
-
Softgels
-
Flow-through cell
显示更多 -
-
Peptide Platform
 显示更多
显示更多 -
Research Team
显示更多
-

Quality Management System
Good Quality Management
Jiuzhou Pharma ensures quality management throughout the entire product lifecycle, from R&D to production, from raw material procurement to product manufacturing and sales, with continuous optimization in every step. We have established quality control and cGMP quality systems that adhere to global industry standards, to ensure the delivery of specialty APIs and CDMO services and meet the rigorous requirements of multinational pharma companies. We have developed graded GMP management systems based on product properties and GMP requirements to ensure product quality while boosting efficiency. These systems provide solid support for our one-stop CDMO services and offer robust quality assurance that secures continuous orders from major multinational pharma companies and promotes steady growth in business performance.
Jiuzhou Pharma's quality management system has been inspected and certified by regulatory agencies in China, Europe, Italy, South Korea, Australia, Brazil, Japan and other countries or regions across the globe to ensure a consistent supply of quality products.
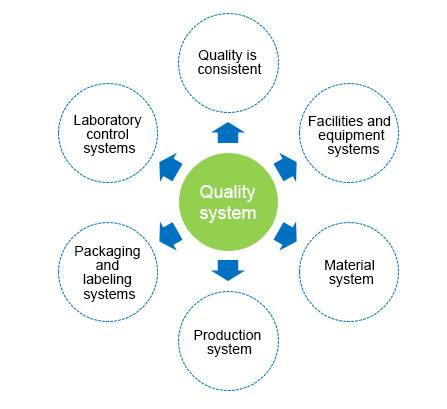
Quality Philosophy
Quality is our lifeblood. We are committed to the values of "Serve Life, Guard Health" throughout our business practices.
Quality Policy
Jiuzhou Pharma pledges that quality is our lifeblood.
We are committed to the values of "Serve Life, Guard Health" throughout our business practices.
We create a rigorous, scientific, and systematic quality assurance system based on risk management by using operations and continuous quality improvement strategies.
We strive relentlessly to meet and exceed customer expectations and regulatory requirements. We are committed to becoming a world-class pharmaceutical company by providing high value-added products and services.
Audit/Certification
Since 2000, Jiuzhou Pharma has passed inspections and certifications by regulatory agencies in China, the USA, Europe, Brazil, Italy, Mexico, and other countries or regions. We also passed quality audits by global customers and earned their approval of our quality system.
10+
Regulatory agencies around the world
70+
Inspections or certifications by regulatory agencies
Inspections or certifications by regulatory agencies around the world (2000—2022):
|
Country/Region
Number of Certifications
|
||
|
China |
|
45+ |
|
USA |
|
7 |
|
Europe |
|
6 |
|
Brazil |
|
6 |
|
Mexico |
|
5 |
|
Italy |
|
3 |
|
Australia |
|
2 |
|
Japan |
|
2 |
|
Hungary |
|
1 |
|
Korea |
|
1 |
Other Quality and Compliance
Business Collaboration
Innovative Drug CDMO Services













